Abstract
Background: Hypomethylating agents (HMAs), such as azacitidine and decitabine, are the current standard of care for patients with myelodysplastic syndrome (MDS). Though HMAs improve cytopenias and delay progression of disease, most patients eventually lose response to these agents and have poor survival afterwards. This poses significant treatment challenges after HMA failure (HMA-F) and an increased risk of transformation to acute myeloid leukemia. The genomic evolution of patients with MDS at the time of HMA-F has not been previously described. Here, we assess the genomic changes of MDS patients at the time of HMA-F and their effects on the outcomes of MDS patients at the time of HMA-F.
Methods: We retrospectively evaluated all patients with MDS seen at a single tertiary cancer center from July 2017 to July 2021 and identified those who were previously untreated and later developed HMA-F. Bone marrow (BM) data was assessed at diagnosis and at the time of HMA-F. Genomic DNA was extracted from whole BM aspirate samples and subject to 81-gene target PCR-based sequencing using a next-generation sequencing platform. Survival data was updated in May 2022. The Kaplan-Meier method and Cox proportional hazards model were used for time-to-event data analysis. The number of metaphases for each unique karyotypic aberration combinations were used to compute the proportions of cytogenetic clones. All the analysis was performed in R 4.0.3.
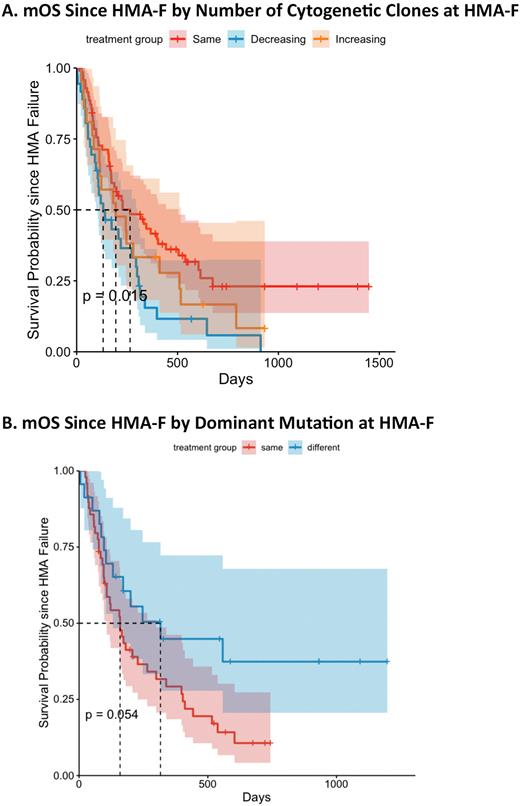
Results: Out of 799 untreated MDS patients, 147 patients (32%) developed HMA-F with a median follow-up time of 44.0 months from diagnosis. Paired cytogenetic and molecular data were available in 128 and 75 patients, respectively. The median overall survival (mOS) since diagnosis (hazard ratio (HR) 1.65, p=0.033) and HMA-F (HR 1.95, p=0.004, Figure A) was inferior for individuals with decreasing number of clones at the time of HMA-F with no statistically significant differences in the time to HMA-F. Gain and/or loss of cytogenetic abnormalities at the time of HMA-F resulted in a shorter mOS (HR 2.35, p<0.001), time to HMA-F (HR 1.65, p=0.033) and mOS since HMA-F (HR 2.89, p<0.001). A change in the dominant cytogenetic clone by the number of metaphases at the time of HMA-F resulted in inferior mOS since diagnosis (HR 2.28, p<0.001), time to HMA-F (HR 1.89, p=0.005), and mOS since HMA-F (HR 2.45, p<0.001). Interestingly, most patients who had gain and/or loss of karyotypic abnormalities and a change in the dominant cytogenetic clone had mutations in TP53. Regarding mutations, there were no statistically significant differences in mOS and time to HMA-F by the gain and/or loss of new mutations at the time of HMA-F. However, the change of the dominant mutation by variant allele frequency resulted in improved mOS from diagnosis (HR 0.47, p=0.02), time to HMA-F (HR 0.46, p=0.02), and mOS since HMA-F (HR 0.55, p=0.072, Figure B). Of note, most TP53-mutated patients had no changes in mutations at the time of HMA-F.
Conclusions: Patients with HMA-F MDS have poorer outcomes with a decreasing number of cytogenetic clones, gain and/or loss of karyotypic aberrations, or a change in the dominant clone by the number of metaphases at the time of HMA-F. However, the gain or loss of mutations on next-generation sequencing appear to have no impact on survival outcomes, though a change in the dominant mutation by variant allele frequency appears to be protective. Further understanding of the underlying biology of HMA-F MDS is warranted with an urgent need for therapeutic interventions after failure of HMA therapy.
Disclosures
Kanagal-Shamanna:Amgen: Consultancy; Novartis: Consultancy; Aptitude Health: Speakers Bureau; Physicians Education Resource: Speakers Bureau. Kadia:cellenkos: Research Funding; Novartis: Consultancy; Pfizer: Research Funding; Genentech: Consultancy, Research Funding; Ascentage: Research Funding; Iterion: Research Funding; JAZZ: Consultancy, Research Funding; Astex: Honoraria; Amgen: Research Funding; PinotBio: Consultancy; AstraZeneca: Research Funding; Genfleet: Research Funding; Delta-Fly: Research Funding; Regeneron: Research Funding; Astellas: Research Funding; cyclacel: Research Funding; Glycomimetics: Research Funding; BMS: Consultancy, Research Funding; Servier: Consultancy; Agios: Consultancy; Abbvie: Consultancy, Research Funding. Jabbour:Spectrum: Research Funding; Pfizer: Other: Advisory Role, Research Funding; AbbVie: Other: Advisory Role, Research Funding; Genentech: Other: Advisory Role, Research Funding; Bristol Myers Squibb: Other: Advisory Role, Research Funding; Amgen: Other: Advisory Role, Research Funding; Adaptive Biotechnologies: Other: Advisory Role, Research Funding; Takeda: Other: Advisory Role, Research Funding. Pemmaraju:stemline: Consultancy; abbvie: Consultancy; immunogen: Consultancy; mustangbio: Research Funding; incyte: Consultancy; novartis: Research Funding; pacylex: Consultancy, Research Funding; samus: Research Funding; daiichi sankyo: Research Funding; cellectis: Research Funding; cellularity: Research Funding. Short:Astellas: Research Funding; Stemline Therapeutics: Research Funding; AstraZeneca: Consultancy; Takeda Oncology: Consultancy, Research Funding; Novartis: Consultancy; Amgen: Consultancy, Honoraria; Pfizer: Consultancy. Ravandi:AstraZeneca: Consultancy; Astex/Taiho: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Amgen: Honoraria, Research Funding; BMS/Celgene: Consultancy, Honoraria, Research Funding; Xencor: Research Funding; Prelude: Research Funding; Novartis: Consultancy; Amgen: Honoraria, Research Funding; Syos: Consultancy, Honoraria, Research Funding; Biomea Fusion, Inc.: Research Funding. Alvarado:FibroGen: Research Funding; Jazz Pharmaceuticals: Research Funding; Astex Pharmaceuticals: Research Funding; Daiichi-Sankyo/Lilly: Research Funding; BerGenBio: Research Funding; Sun Pharma: Research Funding. Colla:Amgen: Research Funding. Kantarjian:Daiichi-Sankyo: Consultancy, Research Funding; Pfizer: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Astellas Health: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Research Funding; Ascentage: Membership on an entity's Board of Directors or advisory committees, Research Funding; NOVA Research: Honoraria; ImmunoGen: Research Funding; Ipsen Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Research Funding; KAHR Medical Ltd: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Honoraria, Research Funding; Takeda: Honoraria. Garcia-Manero:Acceleron Pharma: Consultancy; Curis: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Aprea: Honoraria; Genentech: Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Gilead Sciences: Research Funding; AbbVie: Honoraria, Research Funding; Astex: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.